Each semester, the microbiology students make a visit to my lab to see the scanning electron microscope to get an idea how electron microscopy is done, or at least as much as they soak up in about 20 minutes. During this time they are studying fungi so I use one of the (non-pathogenic) black bread molds as the sample for the SEM, in this case Rhizopus stolonifer. This semester I decided to make a try some new settings on the SEM and was able to make better images than usual so I thought I would show you what I have.
[On a technical note my lab does not have a sputter coater, so I literally use a pair of forceps to transfer a small sample to the copper tape on the stub and put the sample right into the microscope, what I call down and dirty electron microscopy. The SEM in the lab is an Hitachi S-3400N. I used the backscatter detector, working distance 4-5 mm, 70-80 Pascals, 10 kV, probe current 35-50, with the aperture set at 4]
Let's start with the sample. These are cell phone camera images.
 |
| Fungi samples for microbiology are grown in Petri dishes containing potato-dextrose agar. This shows the plate open, lid on the right. The culture was a couple of weeks old and seriously overgrown, but still great for my purposes. |
 |
| A closer look at Rhizopus in the Petri dish. You can see why it is called a black bread mold even though the hyphae - the fibers that make up the "body" of the fungus - are white. The little black spheres here are sporangia. Cumulatively, the sporangia make countless numbers of spores which spread the fungus to new habitats. |
The next two images were taken with a Leica dissecting microscope.
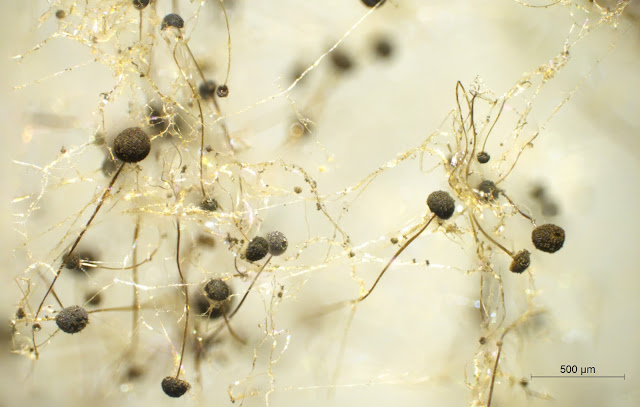 |
| This image is a composite of 28 images that were focus-stacked in Photoshop. It shows the tread-like hyphae and the darker sporangiophores with sporangia at the top. You can just make out the spores that make up the sporangia. |
 |
| Higher magnification of the the same area as shown above. Magnification here is 125x. This is a composite of 17 focus-stacked images. |
So what the heck is focus stacking?
The depth of focus for the lenses on a light microscopes is very shallow which means that if there is any z-axis depth to your sample only one tiny part will be in focus. The image below shows one of the images used to make the previous image and illustrates what I am talking about.
 |
| One of the images used to make the focus-stacked image above. Everything either above or below the focal plane is out of focus. Even the large sporangium at left center is only partially in focus. |
To focus stack you take a series of images, slowing moving the stage down (or up) after each image. Ideally, the focus depths of sequential images should overlap. Once the images are taken, Photoshop does the rest by selecting the part of each image that is in focus and combining those parts into a single image.
As Arthur C. Clarke wrote, "Any sufficiently advanced technology is indistinguishable from magic." I know this is actually very clever programming, but it sure looks "magical" when Photoshop reveals the final image.
Scanning Electron Microscope Images
One of the nice things about electron microscopy is that you don't have to focus stack. The depth of field is much deeper and can be controlled.
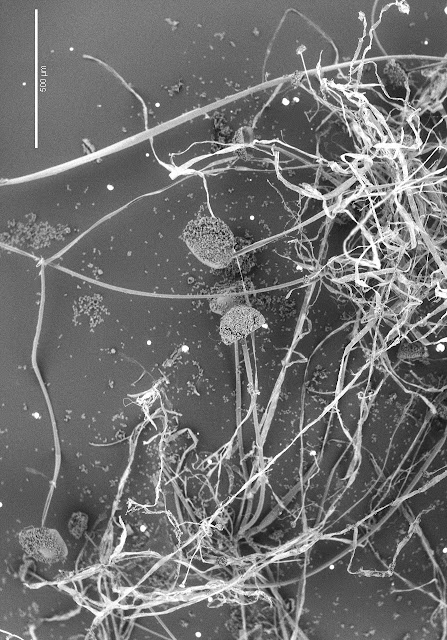 |
| In the image above the white, thread-like filaments that make up the fungus are obvious in the upper, right-hand portion of the image. A mass of hyphae is called a mycelium. [SEM image; 55x] |
 |
| This is the sporangium in the center of the image above. The stalk that supports it is the sporangiophore. [SEM image; 190x] |
 |
| Note that the texture of the spores has become apparent as well as some small crystals of potassium chloride that have formed on the surface of some of the spores. [SEM image; 1,000x] |
 |
| At 5,000x lots of detail shows up in the SEM. |
 |
| Note that the spore in this image has several holes in it. Those were accidentally burned into the spore when I really zoomed in on it. This is called beam damage (dang it). Also notice the details of structure the can be seen in this 15,000x image. |
What causes beam damage and why isn't it across the entire sample?
One trick in electron microscopy is to zoom way, way in - even higher than you plan to capture - and focus. As you back out on the magnification, the entire image remains in focus. The SEM puts out a set amount of energy, regardless of magnification. As you zoom in, the energy is concentrated into a smaller, and smaller area that can, when imaging something like a spore, begin to burn the specimen.
 |
| These holes are actually very tiny. This is a 50,000x magnification image. The SEM is calibrated so I can make measurements directly on the image. This hole is 146 nm across or 0.000146 mm. Small. |
 |
| This image, at 35,000x, shows one of the salt crystals on the surface of the spore. It is 549 nm across. |
 |
| This last image, at 55,100x, is of a salt crystal that was on the side of a spore. I just thought it looked interesting, sort of like the head of a turtle. |
One of the things I try to impress upon our microbiology students is that we humans are in a continuing competition with fungi for our food. You probably don't think about it, but when you throw away old, out-of-date food or food scraps, ultimately, you are feeding the fungi. It is very nice of you go to work every day to make the money to buy them food. I am sure they appreciate it.
~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~

All images in this blog are covered by Creative Commons license. You may download, use, or modify the images any way you like just as long as you attribute the Eastfield College Microscopy Lab and don't sell them.
Best,
Murry Gans










 All images in this blog are covered by Creative Commons license. You may download, use, or modify the images any way you like just as long as you attribute the Eastfield College Microscopy Lab and don't sell them.
All images in this blog are covered by Creative Commons license. You may download, use, or modify the images any way you like just as long as you attribute the Eastfield College Microscopy Lab and don't sell them.